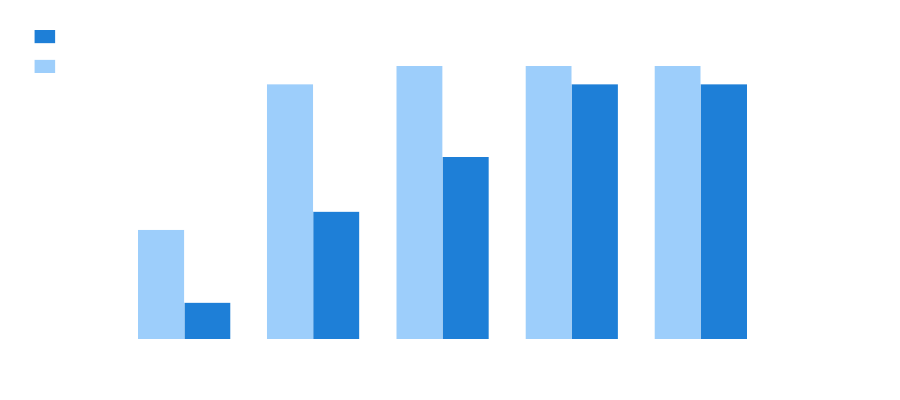In order to improve the registration declaration, experts in Kingfriend have shared their knowledge and experience with staffs in the official accreditation body in China, and constantly summarized experience and improved methods, aiming at achieving a 100% approval rate.
Since 2017, a total of 77 projects have been declared in which 45 have been approved, reaching an approval rate of 56%. Plus, the approval rate is increasing year by year, reaching 94% in 2021.
At present, there over 10 projects being declared, over 10 research projects in progress, and over 10 projects in reserve.